The Basic Concept Behind Nuclear Fission
Radioactive Decay
Before nuclear fission is discussed, a vital scientific concept called radioactive decay must be explained. Radioactive decay is the process by which elements containing unstable nuclei emit energy in the form of electromagnetic waves and subatomic particles. This occurs as a result of the atom's lack of sufficient binding energy to hold the nucleus together. Three basic forms of radioactive decay include, alpha, beta, and gamma decay. These variations are categorized by the energy emitted and matter released. Commonly, this ejection of matter will cause transmutation, or the atom's change of one element into another.
Alpha Decay |
Beta Decay |
Gamma Decay |
|
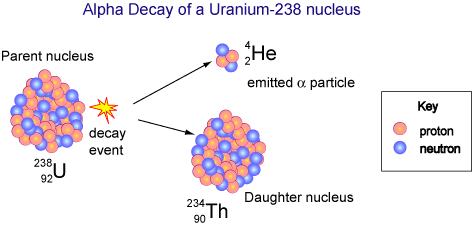
Alpha decay occurs when an unstable, or radioactive, element releases two protons and two neutrons from its nucleus. This conjunction of subatomic particles is called an "alpha particle". As a result, the atom's proton count will drop, therefore forming a new element. This occurs primarily with large, heavier atoms such as uranium or californium.
|
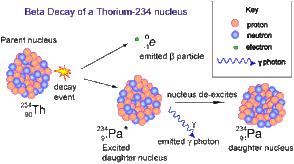
Beta decay is the process by which a nucleus ejects an electron (beta particle), causing a neutron to change into a proton, or vice versa, thus forming a new element.
|
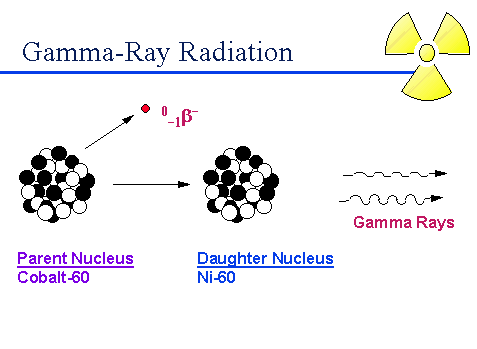
Gamma rays are highly powerful electromagnetic waves with high frequencies. As an element undergoes gamma decay, it will emit gamma radiation along with an alpha or beta particle.
|
Electron Capture |
Spontaneous Fission |
|
Electron capture occurs when an atom or ion passes through a medium and gains a certain number of electrons. Given the atom's disproportionality in charges, the resulting atom will become unstable.
|
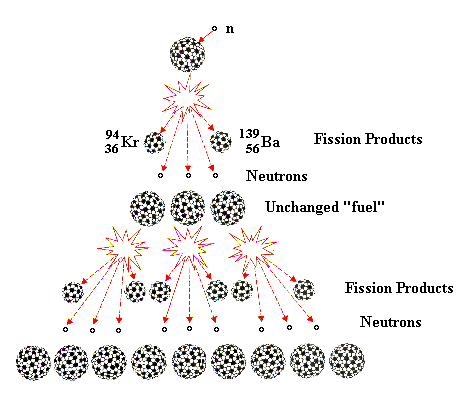
A form of radioactive decay in which a heavier element containing an unstable nucleus splits into two nearly equal fragments by overcoming its binding energy. This will commonly release a neutron in the process.
|
Nuclear Fission and Radioactive Decay
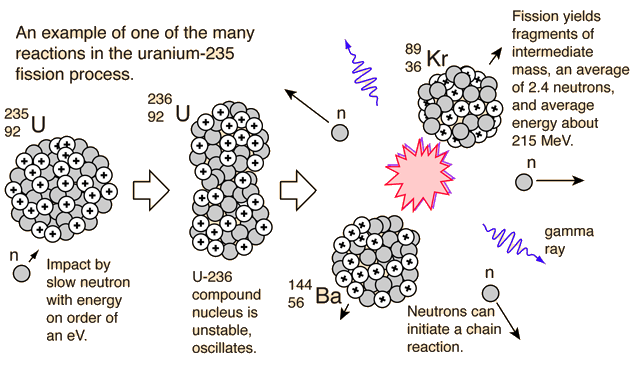
Within a nuclear reactor, an element is able to undergo induced nuclear fission by means of spontaneous fission and radioactive decay. As previously covered, spontaneous fission is the natural release of energy via the atomic "split" of a heavy isotope. Therefore, a neutron will be ejected in the process, and this neutron is able to be captured by the nuclei of other proximal elements. The bombardment of this nearby element will induce instability and thus cause another occurrence of nuclear fission, creating a chain reaction.
Uranium-235: The Nuclear Reactor's Fuel
|
The most commonly utilized isotope is uranium-235, one of the heaviest naturally occurring isotopes. Other variants of uranium, and even plutonium, may be used. This atom is able to undergo induced fission with its natural instability and massiveness. Given its amp amount of neutrons, protons, and electrons, the fragmentation process caused by nuclear fission results in different radioactive isotopic forms of elements such as plutonium, barium, and krypton, elements that are all able to release energy and matter.
Essentially, the binding energy that is overcome in this process directly causes a tremendous amount of energy to be released during the fragmentation of these isotopes. This energy (commonly in the form of gamma radiation) is able to indirectly be converted into electrical energy. To learn specifically how this occurs, visit the "Basics of a Nuclear Reactor" to the right to learn more. Below is a video explanation of nuclear fission.
|